Capsule Evaluation and incompatibility MCQs
GPAT Capsule MCQs on capsule evaluation & incompatibility. Practice AIIMS-level questions on dissolution, disintegration, moisture effects & stability issues
4/27/20266 min read
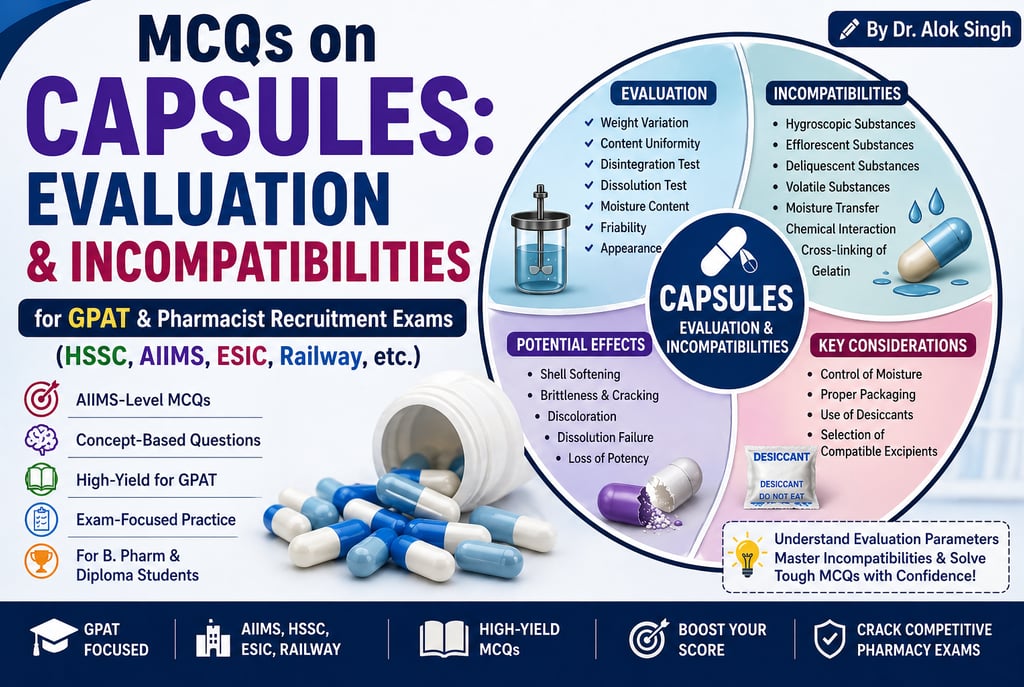
GPAT & Pharmacist Recruitment (HSSC, ESIC, Railway, AIIMS, etc.) MCQ set on Capsules: Evaluation & Incompatibilities.
A. Evaluation of Capsules
1. Which test is used to check the uniformity of weight in capsules?
A. Dissolution test
B. Weight variation test
C. Disintegration test
D. Content uniformity test
Correct Answer: B
2. For capsule evaluation, content uniformity is especially important for:
A. High-dose drugs
B. Low-dose potent drugs
C. Herbal drugs
D. Liquid-filled capsules
Correct Answer: B
3. The disintegration test for capsules is performed using:
A. Friabilator
B. Disintegration apparatus
C. Dissolution tester
D. Hardness tester
Correct Answer: B
4. The medium used for capsule dissolution testing is generally:
A. Distilled water
B. Buffer solutions
C. Alcohol
D. Oil
Correct Answer: B
5. Which parameter ensures the release of drugs from the capsule?
A. Hardness
B. Disintegration
C. Weight variation
D. Friability
Correct Answer: B
6. Which test measures drug release rate from capsules?
A. Disintegration test
B. Dissolution test
C. Weight variation test
D. Moisture test
Correct Answer: B
7. The apparatus used for dissolution testing of capsules is:
A. USP Apparatus I or II
B. Friabilator
C. Hardness tester
D. Viscometer
Correct Answer: A
8. Capsule shells are evaluated for moisture content because
A. It affects color
B. It affects flexibility and brittleness
C. It affects taste
D. It affects size
Correct Answer: B
9. Which test ensures each capsule contains the intended drug amount?
A. Weight variation
B. Content uniformity
C. Disintegration
D. Dissolution
Correct Answer: B
10. Capsules become brittle when:
A. Moisture increases
B. Moisture decreases
C. Temperature increases
D. Pressure increases
Correct Answer: B
B. Incompatibilities in Capsules
11. Which of the following can cause softening of capsule shells?
A. Dry powders
B. Hygroscopic substances
C. Non-polar drugs
D. Lubricants
Correct Answer: B
12. Efflorescent substances in capsules may:
A. Increase moisture
B. Lose water and affect the shell
C. Improve stability
D. Increase hardness
Correct Answer: B
13. Which type of substances absorb moisture and affect the capsule shell?
A. Efflorescent
B. Hygroscopic
C. Hydrophobic
D. Volatile
Correct Answer: B
14. Deliquescent substances cause
A. Drying of the capsule
B. Absorption of moisture leading to liquefaction
C. No effect
D. Hardening of the shell
✅ Correct Answer: B
15. Which incompatibility leads to brittleness of capsule shells?
A. High humidity
B. Low humidity
C. High temperature
D. High pressure
Correct Answer: B
16. Volatile drugs in capsules may:
A. Increase hardness
B. Evaporate and escape
C. Increase moisture
D. Improve dissolution
Correct Answer: B
17. Which of the following substances should NOT be filled directly in capsules without precautions?
A. Lactose
B. Hygroscopic drugs
C. Starch
D. Talc
Correct Answer: B
18. Incompatibility due to moisture exchange can lead to:
A. Improved stability
B. Capsule deformation
C. Increased hardness
D. Better dissolution
Correct Answer: B
19. Which of the following can be used to overcome hygroscopic incompatibility?
A. Adding lubricants
B. Using desiccants
C. Increasing moisture
D. Reducing temperature
Correct Answer: B
20. Cross-linking of gelatin affects:
A. Color
B. Dissolution
C. Size
D. Weight
Correct Answer: B
Most repeated exam points:
Weight variation vs. content uniformity
Disintegration vs Dissolution (very common confusion)
Moisture → brittleness (low) / softening (high)
Hygroscopic & deliquescent → major incompatibility
Cross-linking → ↓ dissolution
Dr. Alok Bains
Assertion–Reason MCQ set on Capsules: Evaluation & Incompatibilities, crafted for GPAT / pharmacist exams.
Assertion–Reason MCQs (Evaluation & Incompatibilities)
Directions:
Choose the correct option:
A. Both Assertion (A) and Reason (R) are true, and R is the correct explanation of A
B. Both A and R are true, but R is NOT the correct explanation of A
C. A is true, but R is false
D. A is false, but R is true
1.
Assertion (A): The dissolution test is more informative than the disintegration test for capsules.
Reason (R): The dissolution test measures the rate and extent of drug release.
Answer: A
2.
Assertion (A): A content uniformity test is essential for low-dose drugs in capsules.
Reason (R): Small variation in drug content can significantly affect therapeutic response.
Answer: A
3.
Assertion (A): Capsules may fail dissolution due to gelatin cross-linking.
Reason (R): Cross-linking reduces the solubility of gelatin shell.
Answer: A
4.
Assertion (A): Capsules become brittle at low humidity.
Reason (R): Loss of moisture reduces the flexibility of the gelatin shell.
Answer: A
5.
Assertion (A): High humidity causes softening of capsule shells.
Reason (R): Excess moisture acts as a plasticizer.
Answer: A
6.
Assertion (A): The weight variation test ensures uniform drug content in capsules.
Reason (R): All capsules contain equal amounts of excipients and drug.
Answer: D
(Weight variation ≠ , drug uniformity, especially for low-dose drugs)
7.
Assertion (A): The disintegration test alone is sufficient to predict drug release from capsules.
Reason (R): Disintegration ensures the breakdown of the capsule shell.
Answer: B
(Both true, but not sufficient → dissolution is needed)
8.
Assertion (A): Hygroscopic drugs can cause capsule shell softening.
Reason (R): They absorb moisture from the environment.
Answer: A
9.
Assertion (A): Efflorescent substances may lead to capsule brittleness.
Reason (R): They lose water of crystallization.
Answer: A
10.
Assertion (A): Deliquescent substances are unsuitable for capsule formulation.
Reason (R): They absorb moisture and may liquefy.
Answer: A
11.
Assertion (A): Volatile drugs may escape from capsule shells.
Reason (R): Capsule shells are permeable to gases.
Answer: A
12.
Assertion (A): Moisture content of the capsule shell must be controlled.
Reason (R): It affects mechanical properties like flexibility and brittleness.
Answer: A
13.
Assertion (A): Dissolution failure can occur even if disintegration is proper.
Reason (R): The drug may have poor solubility.
Answer: A
14.
Assertion (A): Cross-linked gelatin capsules can be tested using enzymes in dissolution media.
Reason (R): Enzymes help break down cross-linked gelatin.
Answer: A
15.
Assertion (A): Capsule shells are impermeable to moisture.
Reason (R): Gelatin forms a tight polymeric network.
Answer: D
16.
Assertion (A): Content uniformity test is not required for high-dose capsules.
Reason (R): The weight variation test is sufficient in such cases.
Answer: A
17.
Assertion (A): Incompatibility due to moisture transfer can affect drug stability.
Reason (R): Moisture can initiate chemical degradation.
Answer: A
18.
Assertion (A): Capsule shells may interact with fill material.
Reason (R): Migration of moisture or components can occur.
Answer: A
19.
Assertion (A): High temperature can accelerate gelatin cross-linking.
Reason (R): It promotes chemical reactions in gelatin.
Answer: A
20.
Assertion (A): Use of desiccants can prevent moisture-related incompatibility.
Reason (R): Desiccants absorb excess environmental moisture.
Answer: A
Points to Think
Distinguish disintegration vs. dissolution (most common trap)
Understand Moisture → Softening vs Brittleness
Remember Cross-linking → Dissolution failure
Identify Incompatibility types (Hygroscopic, efflorescent, and Deliquescent)
Use this quick logic map:
Moisture ↓ → Brittleness
Moisture ↑ → Softening
Cross-linking → ↓ Dissolution
Hygroscopic → absorbs moisture
Efflorescent → loses moisture
Dr. Alok Bain
Difficult, case-based MCQs on Capsules: Evaluation & Incompatibilities. These are designed to test application, integration of concepts, and clinical/industrial reasoningthe
Case 1: Dissolution Failure Despite Proper Disintegration
A pharmaceutical company reports that a batch of hard gelatin capsules disintegrates within the specified time, but fails dissolution testing. Investigation reveals storage at high temperature and humidity.
Question: What is the MOST likely cause?
A. Increase in moisture content
B. Gelatin cross-linking
C. Poor powder flow
D. Capsule size variation
Correct Answer: B
Explanation: Cross-linking reduces gelatin solubility → dissolution failure despite normal disintegration (Most favorite trap)
Case 2: Capsule Brittleness Issue
A batch of capsules becomes brittle and cracks during packaging in winter conditions.
Question: What is the most appropriate corrective measure?
A. Increase plasticizer content
B. Store at low humidity
C. Increase moisture content
D. Add lubricant
Correct Answer: C
Explanation: Low humidity → moisture loss → brittleness → corrected by controlled humidity
Case 3: Softening of Capsules During Storage
Capsules stored in a humid environment become soft and sticky.
Question: Which type of drug is most likely responsible?
A. Hydrophobic drug
B. Hygroscopic drug
C. Efflorescent drug
D. Volatile drug
Correct Answer: B
Case 4: Content Uniformity Failure
A capsule formulation containing a potent drug (low dose) shows acceptable weight variation but fails content uniformity.
Question: What is the most likely issue?
A. Poor mixing of the drug
B. Capsule shell defect
C. High moisture content
D. Poor disintegration
Correct Answer: A
Case 5: Liquefaction Inside Capsule
A formulation contains a deliquescent drug. After storage, the powder inside capsule becomes liquid.
Question: What is the best preventive strategy?
A. Add disintegrant
B. Use desiccant in packaging
C. Increase capsule size
D. Add lubricant
Correct Answer: B
Case 6: Volatile Drug Loss
Capsules filled with a volatile drug show reduced potency over time.
Question: What is the primary reason?
A. Capsule shell degradation
B. Permeability of capsule shell
C. Cross-linking of gelatin
D. Poor filling method
Correct Answer: B
Case 7: Efflorescent Drug Problem
A formulation contains sodium carbonate decahydrate. After storage, capsule shells become brittle.
Question: What is the cause?
A. Absorption of moisture
B. Loss of water of crystallization
C. Chemical degradation
D. Oxidation
Correct Answer: B
Case 8: Dissolution Enhancement Strategy
Capsules fail dissolution due to gelatin cross-linking.
Question: What modification in the test is recommended?
A. Increase temperature
B. Add surfactant
C. Add enzymes to dissolution medium
D. Reduce agitation
Correct Answer: C
Case 9: Poor Flow Powder Filling
A powder blend with poor flow properties is used in capsule filling and shows high weight variation.
Question: Which filling method is most suitable?
A. Dosator method
B. Punch method
C. Tamping pin method
D. Manual filling
Correct Answer: C
Case 10: Capsule Deformation
Capsules stored with hygroscopic excipients show deformation.
Question: What is the primary mechanism?
A. Moisture loss from the shell
B. Moisture absorption by fill material
C. Thermal degradation
D. Mechanical stress
Correct Answer: B
11. Case 11: Disintegration Pass but Bioavailability Low
Capsules pass both disintegration and dissolution tests but show poor bioavailability.
Question: What could be the reason?
A. Poor drug permeability
B. Capsule shell defect
C. High moisture content
D. Low bloom strength
Correct Answer: A
12. Case 12: Capsule Leakage
Liquid-filled capsules show leakage during storage.
Question: What is the best solution?
A. Use enteric coating
B. Apply band sealing
C. Increase plasticizer
D. Reduce moisture
Correct Answer: B
13. Case 13: High Humidity Storage
Capsules stored at high humidity show microbial growth.
Question: What is the most appropriate preventive step?
A. Add lubricant
B. Add preservative
C. Reduce capsule size
D. Increase temperature
Correct Answer: B
14. Case 14: Weight Variation Failure
Capsules filled using the dosator method show variation in weight.
Question: What is the likely cause?
A. Improper vacuum control
B. Excess lubricant
C. High moisture
D. Capsule shell defect
Correct Answer: A
15. Case 15: Capsule Shell Hardening
Capsules become hard and fail to dissolve properly.
Question: What is the most probable cause?
A. Low plasticizer
B. Cross-linking of gelatin
C. High moisture
D. Poor mixing
Correct Answer: B
Learning Takeaways
These cases test:
Cross-linking → dissolution failure
Moisture ↔ shell behavior (softening/brittleness)
Incompatibility types (hygroscopic, efflorescent, deliquescent)
Evaluation logic (weight vs content vs dissolution)
Method selection based on formulation
Train yourself using “Problem → Cause → Solution” mapping:
Dissolution fail → Cross-linking → Add enzyme
Brittleness → Low moisture → Humidity control
Softening → Hygroscopic drug → Desiccant
Dr. Alok Bains.
