IVth Sem DMLT Sample Papers
HSBTE QUESTION SOLUTION
Dr Pramila Singh
4/11/202629 min read
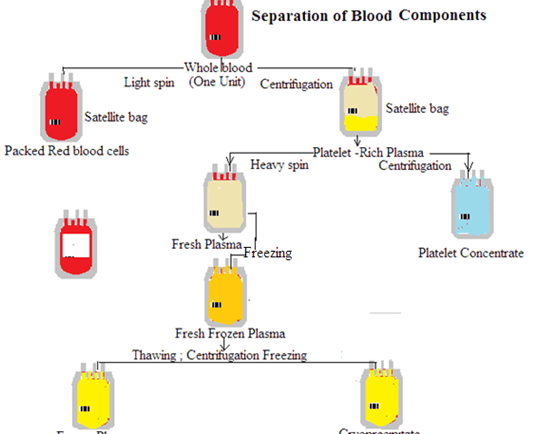
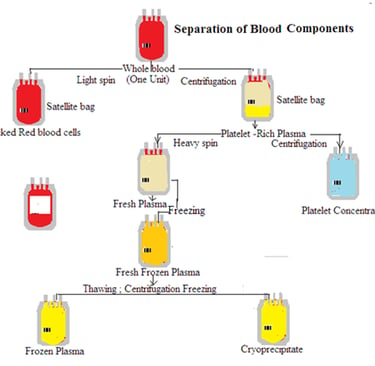
4th Semester/DMLT HSBTE Sample Paper, Dec 25
SET: 1
Subject: Clinical Haematology-II Time : 3 Hrs. 221942 M.M.: 60 221942
SECTION-A Note: Multiple-choice questions. All questions are compulsory (6x1=6)
Q.1 Which of the following cells is primarily involved in the formation of the platelet plug?
a) erythrocytes b) Monocytes c) Platelets d) Neutrophils
Answer: c) Platelets
Platelets adhere to damaged endothelium and aggregate to form the initial plug in hemostasis.
Q.2 Which type of bone marrow is actively involved in blood cell production?
a) Red marrow b) Spongy marrow c) Yellow marrow d) Cortical marrow
Answer: a) Red marrow
Red marrow is hematopoietically active, producing RBCs, WBCs, and platelets. Yellow marrow mainly stores fat.
Q.3 Which test is most definitive for diagnosing leukemia?
a) Peripheral blood smear b) Complete blood count c) Bone marrow biopsy d) ESR test
Answer: c) Bone marrow biopsy
While blood smear and CBC can suggest leukemia, a bone marrow biopsy provides the definitive diagnosis.
Q.4 The LE cell phenomenon is most commonly associated with which disease?
a) Rheumatoid arthritis b) Systemic lupus erythematosus (SLE) c) Scleroderma d) Dermatomyositis
Answer: b) Systemic lupus erythematosus (SLE)
LE (Lupus Erythematosus) cells are characteristic of SLE, caused by neutrophils engulfing antibody-coated nuclear material.
Q.5 What is the normal white blood cell (WBC) count in CSF of a healthy adult?
a) 0-5 cells/ml b) 20-100 cells/ml c) 5-20 cells/ml d) >.100 cells/ml
Answer: a) 0–5 cells/ml
Normal cerebrospinal fluid is nearly acellular, with up to 5 WBCs per microliter considered normal.
Q.6 Which of the following is a normal pH range for semen?
a) 4.0-5.0 b) 5.5-6.0 c) 6.0-7.0 d) 7.2-8.0
Answer: d) 7.2–8.0
Semen is slightly alkaline, which helps neutralize the acidic environment of the vagina to protect sperm.
SECTION-B
Note: Objective/ Completion type questions. All questions are compulsory. (6x1=6)
Q.7 Platelets plays role in ________.
Answer: Hemostasis (blood clotting)
They help stop bleeding by forming a platelet plug and releasing clotting factors.
Q.8 DIC sands for ________.
Answer: Disseminated Intravascular Coagulation
A serious condition where widespread clotting occurs in small vessels, leading to both clotting and bleeding complications.
Q.9 pH of urine is ________
Answer: Normally ranges from 4.5 to 8.0
The average is around 6.0, slightly acidic, but it can vary with diet and health status.
Q.10 Define clot.
Answer: A clot is a semisolid mass formed by blood components (platelets, fibrin, and trapped cells) to stop bleeding and seal injured vessels.
Q.11 Name any two blood disorders.
Anemia
Leukemia
(Other examples: Hemophilia, Thalassemia, Sickle cell disease)
Q.12 What is the role of vitamin K in coagulation?
Answer: Vitamin K is essential for the synthesis of clotting factors II (prothrombin), VII, IX, and X in the liver. Without it, normal blood clotting cannot occur.
SECTION-C
Note: Short-answer type questions. Attempt any eight questions out of the ten questions. (8x4=32)
Q.13 Draw the well-labelled structure of LE Cell.
Q.14 Write a brief note on the intrinsic pathway of coagulation.
Intrinsic Pathway of Blood Coagulation
The intrinsic pathway is one of the two major mechanisms (alongside the extrinsic pathway) that initiate the coagulation cascade, ultimately leading to the formation of a fibrin clot.
Trigger: It is activated when blood comes into contact with negatively charged surfaces (such as exposed collagen in damaged vessels).
Key Steps:
Factor XII (Hageman factor) is activated upon contact with collagen.
Activated Factor XII converts prekallikrein to kallikrein and activates Factor XI.
Factor XIa activates Factor IX.
Factor IXa, with its cofactor Factor VIIIa, calcium ions, and phospholipids, forms the tenase complex.
This complex activates Factor X to Xa, which then enters the common pathway.
Outcome: The intrinsic pathway amplifies clot formation and ensures a stable fibrin clot is produced.
Key Point: Unlike the extrinsic pathway (which is triggered by tissue factor), the intrinsic pathway is entirely contained within the blood. It is slower but more important for clot formation.
Flow Chart
1. Activation of Factor XII: Blood contacts exposed collagen, activating Factor XII (Hageman factor).
2. Conversion of Prekallikrein: Activated Factor XII converts prekallikrein to kallikrein, amplifying the cascade.
3. Activation of Factor XI: Factor XIIa activates Factor XI to Factor XIa.
4. Activation of Factor IX: Factor XIa activates Factor IX to Factor IXa.
5. Formation of Tenase Complex Factor IXa: combines with Factor VIIIa, calcium, and phospholipids to form the tenase complex.
6. Activation of Factor X: The tenase complex activates Factor X to Factor Xa, linking to the common pathway.
Q.15 Give the procedure of bone marrow aspiration.
Step-by-Step Procedure of Bone Marrow Aspiration
1. Preparation
The patient is positioned (commonly lying on their side or stomach).
The most common site is the posterior iliac crest (back of the hip bone); occasionally, the sternum or tibia (in children).
The skin is cleaned thoroughly with an antiseptic solution.
Local anesthesia is administered to numb the area.
2. Insertion of Needle
A special hollow needle (aspiration needle) is inserted through the skin into the bone.
The physician advances the needle carefully into the marrow cavity.
3. Aspiration
A syringe is attached to the needle.
A small amount of liquid bone marrow (about 1–2 ml) is aspirated.
The patient may feel a brief, sharp pain or pressure during aspiration.
4. Sample Collection
The aspirated marrow is placed on slides for microscopic examination.
Additional samples may be collected for specialized tests (cytogenetics, flow cytometry, molecular studies).
5. Post-Procedure Care
The needle is removed, and pressure is applied to stop bleeding.
A sterile dressing is placed over the site.
The patient is advised to keep the area clean and dry for 24 hours.
Key Points
Duration: The procedure usually takes 10–15 minutes.
Risks: Mild pain, bleeding, or bruising at the site; serious complications are rare.
Uses: Diagnosis of leukemia, lymphoma, anemia, myeloma, and monitoring treatment response.
In summary, bone marrow aspiration is a safe, quick, and essential diagnostic test for evaluating blood cell production and detecting hematological diseases.
Q.16 Differentiate between AML and CML.
Feature
AML (Acute Myeloid Leukemia)
CML (Chronic Myeloid Leukemia)
Onset & Progression
AML (Acute Myeloid Leukemia): Rapid onset; symptoms develop within days to weeks
CML (Chronic Myeloid Leukemia): Slow progression; may remain stable for years
Age Group
AML (Acute Myeloid Leukemia): Affects both children and adults
CML (Chronic Myeloid Leukemia): Primarily affects older adults
Cell Type
AML (Acute Myeloid Leukemia): Immature myeloid blast cells accumulate
CML (Chronic Myeloid Leukemia): More mature but abnormal myeloid cells proliferate
Genetic Basis
AML (Acute Myeloid Leukemia): Various mutations (e.g., FLT3, NPM1, IDH1/2)
CML (Chronic Myeloid Leukemia): Almost always associated with the Philadelphia chromosome (BCR-ABL fusion gene)
Classification
AML (Acute Myeloid Leukemia): Classified into subtypes based on genetic changes
CML (Chronic Myeloid Leukemia): Classified into phases: chronic, accelerated, blast crisis
Symptoms
AML (Acute Myeloid Leukemia): Fatigue, bone pain, bruising, infections, shortness of breath, petechiae
CML (Chronic Myeloid Leukemia): Weight loss, night sweats, splenomegaly, sometimes asymptomatic early
Diagnosis
AML (Acute Myeloid Leukemia): Bone marrow biopsy showing >20% blast cells
CML (Chronic Myeloid Leukemia): Blood/marrow tests showing the BCR-ABL gene or Philadelphia chromosome
Q.17 Give a brief account of semen collection and its transportation guidelines.
Semen Collection
Preparation:
The man should abstain from ejaculation for 2–7 days before collection to ensure optimal sample quality.
Hands and the genital area should be washed to avoid contamination.
Collection Method:
Usually obtained by masturbation into a sterile, wide-mouthed container.
Condoms are not recommended unless special non-toxic ones are provided, as regular condoms contain spermicidal agents.
The entire ejaculate must be collected, as the first portion contains the highest concentration of sperm.
Environment:
Collection is ideally done in a clinic’s designated room.
If collected at home, the sample must be delivered quickly under proper conditions.
Transportation Guidelines
Time Frame:
The sample should reach the laboratory within 30–60 minutes of collection.
Temperature:
Must be kept at body temperature (around 37°C) during transport.
Avoid exposure to extreme heat or cold, as this affects sperm motility.
Handling:
The container should be tightly sealed, labeled with patient details, and transported discreetly.
Shaking or vigorous movement should be avoided.
✅ Summary: Proper semen collection and careful transportation are essential to ensure accurate analysis of sperm count, motility, and morphology.
Q.18 Write a short note on synovial fluid.
Synovial Fluid
Definition: Synovial fluid is a clear, viscous fluid found within the cavities of synovial joints.
Composition: It contains water, hyaluronic acid, lubricin, proteins, and small numbers of white blood cells.
Functions:
Lubrication: Reduces friction between articular cartilage during movement.
Shock absorption: Cushions joints against mechanical stress.
Nutrition: Supplies oxygen and nutrients to avascular articular cartilage.
Waste removal: Helps remove metabolic waste from cartilage.
Clinical Importance:
Analysis of synovial fluid is used to diagnose joint disorders such as arthritis, gout, and infections.
Normal fluid is clear and viscous; abnormal fluid may be cloudy, bloody, or less viscous.
✅ Summary: Synovial fluid is essential for smooth, pain-free joint movement and maintaining cartilage health.
Q.19 Differentiate between LE cell and Tart Cell.
LE Cell
Definition: A neutrophil (or sometimes macrophage) that has engulfed denatured nuclear material coated with autoantibodies.
Associated Disease: Characteristic of Systemic Lupus Erythematosus (SLE).
Appearance: Contains a large, homogeneous, smooth, round inclusion (the “LE body”) inside the cytoplasm.
Significance: Diagnostic marker for SLE; represents an autoimmune process.
Tart Cell
Definition: A monocyte or macrophage that has phagocytosed an intact nucleus of another cell.
Associated Disease: Seen in various conditions, not specific to SLE.
Appearance: Contains a whole, unaltered nucleus within its cytoplasm (not homogeneous like LE cell).
Significance: Considered a nonspecific finding; not diagnostic of autoimmune disease.
Key Difference
LE cell: Engulfed denatured nuclear material → specific for SLE.
Tart cell: Engulfed intact nucleus → nonspecific, seen in different conditions.
In short: LE cell = hallmark of lupus; Tart cell = nonspecific phagocytosis of intact nu
Q.20 Write the clinical importance of the prothrombin time index.
Clinical Importance of Prothrombin Time Index
Assessment of Coagulation:
PT index measures the efficiency of the extrinsic and common pathways of coagulation.
It reflects the activity of clotting factors I (fibrinogen), II (prothrombin), V, VII, and X.
Monitoring Anticoagulant Therapy:
Used to monitor patients on warfarin therapy, ensuring the dose is effective but not excessive.
Often expressed as INR (International Normalized Ratio) for standardization.
Detection of Liver Disease:
Since clotting factors are synthesized in the liver, prolonged PT indicates hepatic dysfunction.
Vitamin K Deficiency:
PT index helps detect vitamin K deficiency, which impairs the synthesis of clotting factors.
Bleeding Disorders:
Prolonged PT suggests possible coagulation factor deficiencies or the presence of inhibitors.
✅ Summary: The PT index is a vital test for evaluating clotting function, monitoring anticoagulant therapy, and diagnosing liver disease, vitamin K deficiency, and bleeding disorders.
Q.21 How will you prepare a bone marrow smear?
Preparation of Bone Marrow Smear
Sample Collection:
Obtain bone marrow aspirate using a sterile aspiration needle and syringe.
Collect 1–2 drops of marrow into a clean container or directly onto a glass slide.
Smear Making:
Place a drop of marrow on a glass slide.
Use another slide at a 30–45° angle to spread the drop smoothly across the surface (similar to a blood smear).
Prepare several smears quickly, as marrow clots rapidly.
Drying:
Allow smears to air-dry completely.
Avoid heat, as it may distort cellular morphology.
Staining:
Stain smears with Romanowsky-type stains (e.g., Wright or Giemsa stain).
This highlights different marrow cell types for microscopic examination.
Microscopic Examination:
Examine under the microscope for cell morphology, maturation stages, and abnormal cells.
Key Point: Bone marrow smears are essential for diagnosing hematological disorders like leukemia, anemia, and myeloproliferative diseases.
Q.22 Write a short note on the microscopic examination of CSF.
Microscopic Examination of CSF
Purpose: To evaluate the cellular content of CSF for diagnosing infections, hemorrhage, malignancy, and inflammatory conditions.
Normal Findings:
CSF is normally clear and colorless.
WBC count: 0–5 cells/µL (mostly lymphocytes).
RBCs: absent.
Abnormal Findings:
Increased WBCs: Suggests infection (e.g., bacterial meningitis → neutrophils; viral meningitis → lymphocytes).
Presence of RBCs: Indicates subarachnoid hemorrhage or traumatic tap.
Malignant cells: May be seen in metastatic cancers or CNS leukemia/lymphoma.
Other inclusions: Parasites, fungi, or abnormal plasma cells in specific conditions.
Clinical Importance:
Helps differentiate between bacterial, viral, fungal, and tubercular meningitis.
Detects hemorrhage, malignancy, and autoimmune disorders affecting the CNS.
Summary: Microscopic examination of CSF is crucial for identifying abnormal cells and guiding the diagnosis of infections, hemorrhage, and malignancies.
SECTION-D
Note: Long answer type questions. Attempt any two questions out of the three questions. (2x8=16)
Q.23 Give the principle, procedure, and normal value of clotting time.
Q.24 Discuss various pathways of coagulation.
Q.25 Write the FAB classification of leukemias.
4th Semester/DMLT, DMLT Set 1
Subject : Immunopathology & Cytology 221946 Dec 25 Time : 3 Hrs. M.M.: 60
SECTION-A
Note: Multiple choice questions. All questions are compulsory (6x1=6)
Q.1 Which stain is used to demonstrate iron in tissues?
a) Giemsa b) Prussian blue c) Sudan III d) PAS
Answer: b) Prussian blue
Prussian blue stain highlights ferric iron deposits (hemosiderin) in tissues.
Q.2 What is the most commonly used laxative in histopathology?
a) Ethanol b) Glutaraldehyde
b) Formalin(10% neutral buffered formalin) d) Acetone
Answer: c) Formalin (10% neutral buffered formalin)
Formalin is the standard fixative for preserving tissue morphology.
Q.3 Which of the following is not typically required for the FNAC procedure
a) Syringe b) Glass slide c) Local anesthesia d) Needle
Answer: c) Local anesthesia
FNAC (Fine Needle Aspiration Cytology) usually requires only a needle, syringe, and slides. Local anesthesia is not routinely used.
Q.4 Which stain is used to demonstrate mucin in cytological smears?
a) Ziehl-Neelsen b) Periodic acid-Schiff(PAS) c) Giemsa d) Masson's trichrome
Answer: b) Periodic acid-Schiff (PAS)
PAS stain detects mucopolysaccharides and mucin substances.
Q.5 Reticulin stain is used to demonstrate:
a) Elastic fibers b) Nerve endings c) Reticular fibers (type III collagen) d) Muscle fibers
Answer: c) Reticular fibers (type III collagen)
Reticulin stain highlights the delicate reticular fibers in tissues.
Q.6 Which of the following is not a component of Kaiserling's solution?
a) Formalin b) Sodium chloride c) Potassium nitrate d) Ethanol
Answer: d) Ethanol
Kaiserling’s solution contains formalin, sodium chloride, and potassium nitrate, but not ethanol
SECTION-B
Note: Objective/ Completion type questions. All questions are compulsory. (6x1=6)
Q.7 The aim of fixation is to cells and tissue constituents. Oil red O stain is used for _________ staining.
Fixation aims to preserve cells and tissue constituents. Oil Red O stain is used for lipid/fat staining.
Q.8 Name any two fixatives.
Examples: Formalin and Glutaraldehyde.
Q.9 Autopsy procedure is used for external examination and examination.
The autopsy procedure is used for external examination and internal examination.
Q.10 method is used to take a sample from the superficial mass using a sharp needle.
Fine Needle Aspiration Cytology (FNAC) method.
Q.11 HCG test is done for
Pregnancy detection (Human Chorionic Gonadotropin hormone is elevated in pregnancy).
SECTION-C
Note: Short answer type questions. Attempt any eight questions out often questions. (8x4=32)
Q.12 Write the principle of the ZN staining method.
The principle of the Ziehl–Neelsen (ZN) staining method is based on the property of acid-fastness. Certain bacteria, especially Mycobacterium tuberculosis, have cell walls rich in mycolic acids and waxy lipids. These lipids make them resistant to ordinary stains but allow them to retain the primary stain even after treatment with strong acid-alcohol.
In summary, the ZN method works because acid-fast bacteria resist decolorization due to their lipid-rich cell walls, allowing them to be distinguished from non–acid-fast organisms.
Q.13 Give the properties of ideal decalcification agents.
The properties of ideal decalcification agents in histopathology:
Rapid action: Should remove calcium quickly without delaying tissue processing.
Preserve tissue morphology: Must not damage or distort cellular and structural details.
Preserve staining qualities: Should not interfere with subsequent staining procedures (H&E, special stains).
Non‑destructive to nucleic acids and proteins: Should allow molecular studies (DNA/RNA/protein analysis) if needed.
Minimal shrinkage or swelling: Should maintain tissue size and architecture.
Easy penetration: Should diffuse uniformly into the tissue for complete decalcification.
Stable and safe: Should be chemically stable, non‑toxic, and easy to handle.
Compatible with fixatives: Should work well with common fixatives like formalin.
Q.14 Write a short note on care and maintenance of the freezing microtome.
Care and Maintenance of Freezing Microtome
Cleaning after use: Always remove tissue debris and frost from the stage and knife holder immediately after sectioning.
Lubrication: Apply a thin layer of suitable lubricant to moving parts regularly to ensure smooth operation.
Knife care: Keep the microtome knife sharp, clean, and free from rust. Store it in a protective case when not in use.
Temperature control: Ensure the freezing stage is maintained at the correct temperature; avoid excessive frost formation.
Avoid corrosion: Wipe down metallic parts to prevent rusting, especially after exposure to moisture.
Calibration: Periodically check and adjust the thickness setting mechanism for accuracy.
Safe handling: Handle the knife and moving parts carefully to prevent accidents and damage.
Storage: Cover the instrument when not in use to protect it from dust and environmental damage.
Q.15 What are the uses of an autopsy?
Uses of Autopsy
Determine cause of death: Establishes the medical reason for death, especially in sudden or unexplained cases.
Study disease processes: Provides valuable information about the progression and effects of diseases on organs and tissues.
Detect medical errors: Helps identify diagnostic mistakes or treatment complications.
Legal and forensic purposes: Assists in criminal investigations, confirming or ruling out foul play.
Public health insights: Contributes to understanding epidemics, new diseases, or environmental hazards.
Medical education and research: Serves as a teaching tool for students and a source of data for scientific studies.
Family reassurance: Offers closure and answers to relatives regarding the cause of death.
Q.16 Write the composition of the Keiserling II solution.
Composition of Kaiserling II Solution
Formalin (37–40% formaldehyde) – 200 mL
Potassium nitrate – 15 g
Potassium acetate – 30 g
Distilled water – up to 1000 mL
Notes
Kaiserling II solution is part of the Kaiserling’s three-solution method for preserving tissues with natural color.
Kaiserling I is the fixative, Kaiserling II is the preservative, and Kaiserling III is the storage solution.
The potassium salts help maintain tissue transparency and color stability.
Q.17 Briefly discuss the FNAC method with a diagram.
FNAC Method – Brief Discussion
Principle: FNAC involves using a fine, hollow needle to aspirate cellular material from a superficial or deep-seated lesion for cytological examination.
Procedure:
A fine needle (usually 22–25 gauge) attached to a syringe is inserted into the lesion.
Negative pressure is applied by pulling back the syringe plunger to aspirate cells.
The material is expelled onto a glass slide, smeared, and stained (commonly with Giemsa or Papanicolaou stain).
Microscopic examination is performed to assess cellular morphology.
Uses: Quick, minimally invasive, cost-effective diagnostic tool for tumors, cysts, lymph nodes, thyroid, breast lumps, etc.
Advantages: Simple, outpatient procedure, avoids surgical biopsy, and rapid diagnosis.
Simple Diagram (conceptual description)
[ Lesion/Mass ]
|
---- Needle ----> Connected to Syringe
|
Aspiration of cells
|
Expelled onto the Glass Slide
|
Smear → Staining → Microscopy
This diagram shows the flow: lesion → needle aspiration → slide preparation → staining → microscopic diagnosis.
Q.18 Write a short note on automation in cytology.
Automation in Cytology
Definition: Automation in cytology refers to the use of advanced instruments, computer-assisted systems, and digital technologies to improve the efficiency, accuracy, and reproducibility of cytological examinations.
Key Features:
Automated slide preparation: Machines prepare smears, fix, and stain slides uniformly.
Automated staining: Ensures consistency in special stains (e.g., Papanicolaou stain).
Image analysis systems: Computer-assisted microscopes scan slides and highlight suspicious cells.
Liquid-based cytology (LBC): Automated systems process samples to reduce artifacts and improve cell preservation.
Digital cytology: High-resolution scanners digitize slides for remote viewing and telepathology.
Advantages:
Reduces human error and variability.
Speeds up processing and reporting.
Improves detection of abnormal cells (e.g., precancerous lesions in cervical cytology).
Facilitates large-scale screening programs.
Limitations:
High cost of equipment.
Requires technical expertise and maintenance.
Still needs human oversight for final diagnosis.
Automation has revolutionized cytology by combining precision, speed, and digital innovation, making diagnostic processes more reliable and scalable.
Q.19 Explain the procedure of a pregnancy test.
A pregnancy test works by detecting the hormone hCG (human chorionic gonadotropin) in urine or blood, which is produced after implantation of a fertilized egg. The test is simple, rapid, and widely used to confirm pregnancy.
Principle
Human Chorionic Gonadotropin (hCG) is secreted by trophoblastic cells of the placenta after implantation.
Pregnancy tests detect hCG either qualitatively (positive/negative) or quantitatively (exact concentration).
hCG can be detected in urine (home test kits) or blood (laboratory tests).
Procedure
1. Urine Pregnancy Test (Home Test Kits)
Collect a fresh urine sample (preferably early morning urine for higher hCG concentration).
Place a few drops on the test strip or dip the strip into the urine.
Wait for 3–5 minutes.
Interpretation:
One line = control (test valid, negative result).
Two lines = control + test line (positive result, hCG present).
2. Laboratory Urine Test
Uses latex particles coated with anti-hCG antibodies.
If hCG is present, agglutination occurs, confirming pregnancy.
3. Blood Test
More sensitive; can detect hCG levels as low as 1 mIU/mL.
Performed in a lab using immunoassays.
Provides quantitative results to monitor pregnancy progression or detect abnormalities.
Key Points
Best time to test: About 1 week after a missed period for reliable results.
False negatives: Can occur if the test is done too early, before hCG levels rise sufficiently.
Clinical use: Besides pregnancy detection, hCG tests are also used in diagnosing ectopic pregnancy, monitoring trophoblastic disease, and certain tumors.
In short, Pregnancy tests detect hCG in urine or blood. Home kits provide quick qualitative results, while lab tests provide more sensitive, quantitative measurements.
Q.20 How will you preserve museum specimens?
Preservation of Museum Specimens
Fixation: Specimens are first fixed in formalin (10% neutral buffered formalin) to prevent autolysis and putrefaction.
Decalcification (if needed): Hard tissues like bone are decalcified using agents such as nitric acid or EDTA.
Kaiserling’s method: A widely used technique for museum specimens. It involves three solutions:
Kaiserling I (formalin, potassium nitrate, potassium acetate) – for fixation.
Kaiserling II – for preservation and maintaining natural color.
Kaiserling III – for long-term storage.
Mounting: Specimens are placed in glass jars filled with preservative solution, ensuring complete immersion.
Labeling: Each specimen is labeled with details like name, diagnosis, and accession number.
Maintenance: Regular checking of fluid levels, topping up with preservative, and replacing cloudy solutions to prevent deterioration.
In short, Museum specimens are preserved by fixation, storage in preservative solutions (such as Kaiserling’s), proper mounting, and regular maintenance to retain their natural appearance and educational value.
Q.21 Explain the process of frozen section cutting.
Specimen Collection
Fresh tissue is obtained during surgery for immediate examination.
The surgeon provides a small tissue sample
No prior fixation is done
Tissue must be kept moist
Freezing the Tissue
The specimen is rapidly frozen to make it firm for sectioning.
Place tissue on the freezing stage of the cryostat
Use liquid CO₂ or refrigerant to freeze
Ensure complete freezing to avoid tearing
Section Cutting
Thin slices of frozen tissue are cut using a freezing microtome/cryostat.
Adjust thickness (usually 5–10 µm)
Cut sections carefully to avoid folds
Transfer sections onto glass slides
Staining the Section
Sections are quickly stained for microscopic examination.
Common stains: Hematoxylin & Eosin (H&E)
Rapid staining protocols were used
Mount with a coverslip
Microscopic Examination
A pathologist examines stained sections to provide an immediate diagnosis.
Check for tumor margins
Identify tissue type and pathology
Report findings to the surgeon
SECTION-D
Note: Long answer type questions. Attempt any two questions out of the three questions. (2x8=16)
Q.23 Give the principle and procedure of the AFB staining method.
Q.24 Describe the principle and procedure of the H & E staining method.
Q.25 “Explain the procedure of autopsy examination
Dr Pramila Sigh
4th Semester/DMLT, Subject: Medical Laboratory Management 221947 Dec 25
Time : 3 Hrs. M.M.: 60
SECTION-A Note: Multiple-choice questions. All questions are compulsory (6x1=6)
Q.1 The example of clinical laboratory equipment used to separate particles by centrifugal force
a) Centrifuge d) Microscope c) Microtome b) All the above
Answer: a) Centrifuge
Centrifuge → It is the clinical laboratory equipment used to separate particles by centrifugal force.
Q.2 The inventory management principle is/are:
a) Minimize companies coast b) Maximize its return c) Both d) None of these
Answer: c) Both
The inventory management principle is to minimize cost and maximize return.
Q.3 CHCOOH is an example of an:
a) Alkali b) Acid c) Neutral d) All of these
Answer: b) Acid
CH₃COOH (acetic acid) is an example of an acid.
Q.4 Examples of biological waste are:
a) Clinical specimen b) Cultures c) Human body fluid samples d) All the above
Answer: d) All the above
Biological waste includes clinical specimens, cultures, and human body fluid samples.
Q.5 Malpractice insurance covers are covered under the act:
a) NABL b) NABH c) ISO d) CPA
Answer: d) CPA
Malpractice insurance is covered under the CPA (Consumer Protection Act).
Q.6 Plastic wares are air dried at a temperature______.
a) 370 b) 60-700 c) 500 d) 1000
Answer: b) 60–700
Plastic wares are air-dried at a temperature of 60–70°C.
SECTION-B
Note: Objective/ Completion type questions. All questions are compulsory. (6x1=6)
Q.7 Define personal protective equipment.
Personal protective equipment refers to specialized clothing or gear (like gloves, masks, gowns, goggles, and face shields) designed to protect healthcare workers and laboratory staff from exposure to infectious agents, chemicals, or physical hazards.
Q.8 Define procurement
Procurement is the process of acquiring goods, services, or equipment from external suppliers. It ensures that the right items are purchased at the right time, quality, and cost to meet organizational needs.
Q.9 Enlist 4 lab equipment.
Centrifuge, Microscope, Autoclave, Spectrophotometer
Q.10 Write any two examples of accreditation bodies in India for clinical laboratories.
NABL (National Accreditation Board for Testing and Calibration Laboratories)
NABH (National Accreditation Board for Hospitals & Healthcare Providers)
Q.11 Define transportation.
Transportation in a laboratory or healthcare context refers to the safe and secure movement of specimens, samples, or medical materials from one location to another. It ensures integrity and prevents contamination or damage.
Q.12 Define retrieval of data.
Retrieval of data is the process of accessing and extracting stored information from databases, records, or archives for analysis, reporting, or decision-making.
SECTION-C
Note: Short-answer type questions. Attempt any eight questions out of the ten questions. (8x4=32)
Q.13 Write the objectives of laboratory layouts and designs.
Objectives of Laboratory Layouts and Designs
Safety Assurance
Efficient Workflow
Space Utilization
Segregation of Activities
Accessibility and Convenience
Compliance with Standards
Ergonomics and Comfort
Data and Resource Management
Q.14 Explain the importance of the disposal of biomedical waste.
Importance of Biomedical Waste Disposal
Prevents the spread of Infection
Protects Public Health
Environmental Protection
Legal and Regulatory Compliance
Safety of Healthcare Workers
Promotes Community Confidence
Resource Management
Q.15 Describe the different financial resources for funding of clinical laboratories.
Financial Resources for Clinical Laboratory Funding
1. Government Grants & Public Funding
ICMR (Indian Council of Medical Research): Provides intramural and extramural grants for biomedical and public health research.
Department of Biotechnology (DBT) and Department of Science & Technology (DST): Offer funding for laboratory infrastructure and innovation projects.
National Health Mission (NHM): Supports diagnostic facilities in public health labs.
2. Private Investments & Partnerships
Venture capital and angel investors fund for diagnostic startups and private labs.
Corporate partnerships with hospitals or pharmaceutical companies provide financial backing for specialized testing facilities.
3. Loans & Credit Facilities
Commercial banks and financial institutions extend business loans for laboratory setup and expansion.
Specialized healthcare financing schemes are available for equipment purchase and working capital.
4. Accreditation-Linked Support
Laboratories accredited by NABL or NABH often gain easier access to government contracts and private collaborations.
Accreditation enhances credibility, attracting more clients and funding opportunities.
5. International & Non-Governmental Funding
WHO, UNICEF, and other global health organizations provide grants for labs working on infectious disease control.
NGOs and charitable foundations support diagnostic services in underserved regions.
6. Revenue from Services
Clinical labs generate internal funding through patient testing fees, corporate health packages, and hospital tie-ups.
Subscription-based diagnostic services and preventive health check-up programs also contribute to a steady income.
Q.16 Differentiate between pre-analytical and post-analytical factors of quality assurance.
Pre-Analytical Factors (Before Testing)
These are steps that occur before the actual laboratory analysis. Errors here can compromise the accuracy of results.
Patient Preparation: Ensuring fasting, medication restrictions, or proper hydration before sample collection.
Specimen Collection: Correct technique, appropriate container, and proper labeling.
Specimen Handling & Transport: Maintaining temperature, avoiding delays, and preventing contamination.
Data Entry & Identification: Accurate patient information and test requisition forms.
Common Errors: Mislabeling, hemolyzed samples, wrong anticoagulant, or delayed transport.
Post-Analytical Factors (After Testing)
These involve processes after the laboratory analysis is complete, ensuring results are communicated and used correctly.
Result Validation: Checking accuracy, consistency, and plausibility of test results before release.
Report Preparation: Clear, standardized formatting of laboratory reports.
Result Communication: Timely delivery to clinicians or patients, using secure channels.
Interpretation & Clinical Use: Ensuring clinicians understand the results in the right context.
Archiving & Data Retrieval: Proper storage of results for future reference and audits.
Common Errors: Delayed reporting, transcription mistakes, misinterpretation, or loss of records.
Q.17 Write any four principles to be followed in the management of a clinical laboratory.
Principles of Clinical Laboratory Management
Quality Assurance and Accuracy
Ensure all laboratory tests are performed with precision and reliability.
Implement standard operating procedures (SOPs) and regular audits to maintain consistency.
Safety and Compliance
Follow biosafety guidelines to protect staff and patients.
Comply with national and international regulations (e.g., NABL, ISO standards).
Efficient Resource Utilization
Optimize use of manpower, equipment, and consumables.
Maintain proper inventory management to avoid shortages or wastage.
Effective Data and Record Management
Use computerized systems for patient records, test results, and reporting.
Ensure confidentiality, secure storage, and easy retrieval of data.
Why These Principles Matter
They guarantee accurate diagnostic results.
They protect healthcare workers and patients.
They ensure cost-effectiveness and sustainability.
They build trust and credibility for the laboratory.
Q.18 Explain the care and maintenance of the centrifuge used in a clinical laboratory.
1 Regular Cleaning
Safety
Keep the centrifuge free from dust, spills, and biological contaminants.
Wipe rotor chamber and exterior with mild detergent
Disinfect surfaces after handling biological samples
Avoid harsh chemicals that damage plastic or metal parts
2 Rotor Care
Rotors are critical components that must be handled carefully.
Inspect for cracks or corrosion regularly
Clean with non-abrasive solutions
Store rotors in a dry, cool place when not in use
3 Balance Samples
Proper balancing prevents mechanical stress and accidents.
Always load tubes of equal weight opposite each other
Use balance tubes with water if the sample volume is uneven
Check alignment before starting
4 Temperature and Speed Monitoring
Ensure the centrifuge operates within recommended limits.
Do not exceed manufacturer’s speed ratings
Monitor temperature for refrigerated centrifuges
Allow cooling periods between runs
5 Routine Inspection
Regular checks prevent breakdowns and ensure safety.
Verify lid lock and safety interlocks
Check electrical connections and display panels
Schedule professional servicing annually
6 Proper Storage
Store the centrifuge correctly when not in use.
Keep in a clean, dry environment
Cover with dust-protective lid or cloth
Avoid exposure to direct sunlight or moisture
Q.19 Write a detailed note on the code of conduct of members of the medical profession.
Code of Conduct of Medical Professionals
The code of conduct provides ethical and professional guidelines that doctors and healthcare workers must follow to ensure patient welfare, integrity, and trust in the medical profession.
1. Patient-Centered Care
Always prioritize the health, safety, and dignity of patients.
Provide care without discrimination based on race, gender, religion, or financial status.
2. Confidentiality
Protect patient information and medical records.
Share details only with authorized personnel or when legally required.
3. Informed Consent
Ensure patients understand the nature, risks, and benefits of procedures before they agree.
Respect the patient’s autonomy and right to refuse treatment.
4. Professional Competence
Maintain up-to-date knowledge and skills through continuous medical education.
Avoid practices beyond one’s expertise.
5. Integrity and Honesty
Provide truthful information to patients and colleagues.
Avoid false claims, misleading advertisements, or unethical financial practices.
6. Avoidance of Malpractice
Do not engage in negligence, exploitation, or misconduct.
Ensure safe and evidence-based medical practices.
7. Respect for Colleagues
Maintain professional relationships with peers.
Avoid defamation or unfair criticism of fellow practitioners.
8. Social Responsibility
Contribute to public health awareness and preventive medicine.
Participate in community health programs and uphold societal trust.
9. Legal and Ethical Compliance
Follow national laws, medical council regulations, and accreditation standards.
Uphold the reputation of the medical profession.
Importance
Builds trust between patients and doctors.
Ensures quality healthcare delivery.
Protects patients from exploitation and harm.
Maintains the honor and dignity of the medical profession.
Q.20 Define medical ethics and write its importance.
Definition of Medical Ethics
Medical ethics refers to the set of moral principles, values, and professional standards that guide healthcare professionals in their practice. It ensures that patient care is delivered with respect, fairness, and integrity, while protecting the rights and dignity of individuals.
Importance of Medical Ethics
Protects Patient Rights
Ensures confidentiality, informed consent, and respect for autonomy.
Safeguards patients from exploitation or harm.
Promotes Trust
Builds confidence between patients and healthcare providers.
Encourages transparency and honesty in medical practice.
Guides Professional Conduct
Provides a framework for decision-making in complex situations.
Helps doctors and lab professionals balance patient welfare with scientific advancement.
Ensures Fairness and Justice
Promotes equal treatment regardless of background, gender, or financial status.
Prevents discrimination in healthcare delivery.
Supports Quality of Care
Encourages accountability and responsibility in clinical practice.
Improves patient safety and overall healthcare standards.
Legal and Accreditation Compliance
Aligns medical practice with laws, regulations, and accreditation requirements.
Reduces risk of malpractice and legal disputes.
Summary
Medical ethics is the backbone of healthcare practice. It ensures that medical professionals act responsibly, prioritize patient welfare, and maintain integrity in all aspects of clinical care.
Q.21 Write a short note on the uses of computers for various lab services
Uses of Computers in Clinical Laboratories
Data Management
Store, organize, and retrieve patient records, test results, and laboratory reports.
Reduce paperwork and improve accuracy in documentation.
Quality Assurance
Monitor pre-analytical, analytical, and post-analytical processes.
Track errors, maintain compliance, and support accreditation standards.
Instrument Control
Interface with automated analyzers, spectrophotometers, and centrifuges for precise operation.
Enable calibration, monitoring, and troubleshooting of equipment.
Inventory Management
Track reagents, consumables, and plasticware.
Generate alerts for stock shortages and expiry dates.
Communication & Reporting
Generate electronic laboratory reports.
Share results securely with clinicians, hospitals, and patients.
Research & Analysis
Perform statistical analysis of test data.
Support biomedical research through modeling, simulations, and bioinformatics.
Billing & Administration
Automate billing, payment records, and financial management.
Integrate with hospital information systems for smooth workflow.
Importance
Computers enhance efficiency, accuracy, safety, and speed in laboratory services, making them indispensable for modern clinical labs.
Q.22 Explain how the plasticware is maintained in the clinical laboratory.
Maintenance of Plasticware in Clinical Laboratories
Cleaning
Wash immediately after use with mild detergent and warm water.
Avoid strong abrasives or harsh chemicals that can scratch or degrade plastic surfaces.
Rinse thoroughly with distilled water to remove detergent residues.
Disinfection
Use appropriate disinfectants (e.g., 70% ethanol, sodium hypochlorite) depending on the type of contamination.
Ensure contact time is sufficient for effective microbial kill.
Avoid prolonged exposure to strong oxidizing agents that may damage plastic.
Sterilization
Many plastic items cannot withstand autoclaving at high temperatures.
Instead, sterilize using ethylene oxide gas, UV radiation, or chemical sterilants when required.
Disposable plasticware should be used for critical sterile applications.
Drying
Air-dry plasticware at controlled temperatures (commonly 60–70°C) to prevent warping or melting.
Avoid direct sunlight or excessive heat exposure.
Storage
Store in clean, dust-free cabinets or sealed containers.
Keep away from direct sunlight and high humidity to prevent degradation.
Segregate sterile and non-sterile plasticware.
Handling
Handle with care to avoid scratches, cracks, or deformation.
Use designated racks or holders for pipettes, tubes, and flasks.
Avoid mixing plasticware with glassware during washing to prevent damage.
Inspection & Replacement
Regularly check for cracks, discoloration, or deformation.
Replace damaged or worn-out plasticware immediately to maintain accuracy and safety.
SECTION-D
Note: Long answer type questions. Attempt any two questions out of the three questions. (2x8=16)
Q.23 Draw a layout of a clinical laboratory and write the functions of the clinical laboratory in total healthcare.
Q.24 Explain various safety measures in the clinical Laboratory.
Q.25
a) Describe the care and maintenance of the water bath.
b) Explain the role of the First Aid Kit in case of an electric shock in a clinical Laboratory.
Dr. Pramila Singh
4th Semester/DMLT,
Subject: Immunopathology & Cytology 221946 Dec 25 Time : 3 Hrs. M.M.: 60
SECTION-A
Note: Multiple choice questions. All questions are compulsory (6x1=6)
Q.1 Which stain is used to demonstrate iron in tissues?
a) Giemsa b) Prussian blue c) Sudan III d) PAS
Answer: b) Prussian blue
Prussian blue stain highlights ferric iron deposits (hemosiderin) in tissues.
Q.2 What is the most commonly used laxative in histopathology?
a) Ethanol b) Glutaraldehyde
b) Formalin(10% neutral buffered formalin) d) Acetone
Answer: c) Formalin (10% neutral buffered formalin)
Formalin is the standard fixative for preserving tissue morphology.
Q.3 Which of the following is not typically required for the FNAC procedure
a) Syringe b) Glass slide c) Local anesthesia d) Needle
Answer: c) Local anesthesia
FNAC (Fine Needle Aspiration Cytology) usually requires only a needle, syringe, and slides. Local anesthesia is not routinely used.
Q.4 Which stain is used to demonstrate mucin in cytological smears?
a) Ziehl-Neelsen b) Periodic acid-Schiff(PAS) c) Giemsa d) Masson's trichrome
Answer: b) Periodic acid-Schiff (PAS)
PAS stain detects mucopolysaccharides and mucin substances.
Q.5 Reticulin stain is used to demonstrate:
a) Elastic fibers b) Nerve endings c) Reticular fibers (type III collagen) d) Muscle fibers
Answer: c) Reticular fibers (type III collagen)
Reticulin stain highlights the delicate reticular fibers in tissues.
Q.6 Which of the following is not a component of Kaiserling's solution?
a) Formalin b) Sodium chloride c) Potassium nitrate d) Ethanol
Answer: d) Ethanol
Kaiserling’s solution contains formalin, sodium chloride, and potassium nitrate, but not ethanol
SECTION-B
Note: Objective/ Completion type questions. All questions are compulsory. (6x1=6)
Q.7 fixation aims to preserve cells and tissue constituents. Oil red O stain is used for _________ staining.
Fixation aims to preserve cells and tissue constituents. Oil Red O stain is used for lipid/fat staining.
Q.8 Name any two fixatives.
Examples: Formalin and Glutaraldehyde.
Q.9 Autopsy procedure is used for external examination and______________ examination.
The autopsy procedure is used for external examination and internal examination.
Q.10 method is used to take a sample from the superficial mass using a sharp needle.
Fine Needle Aspiration Cytology (FNAC) method.
Q.11 HCG test is done for
Pregnancy detection (Human Chorionic Gonadotropin hormone is elevated in pregnancy).
SECTION-C
Note: Short answer type questions. Attempt any eight questions out often questions. (8x4=32)
Q.12 Write the principle of the ZN staining method.
The principle of the Ziehl–Neelsen (ZN) staining method is based on the property of acid-fastness. Certain bacteria, especially Mycobacterium tuberculosis, have cell walls rich in mycolic acids and waxy lipids. These lipids make them resistant to ordinary stains but allow them to retain the primary stain even after treatment with strong acid-alcohol.
In summary, the ZN method works because acid-fast bacteria resist decolorization due to their lipid-rich cell walls, allowing them to be distinguished from non–acid-fast organisms.
Q.13 Give the properties of ideal decalcification agents.
The properties of ideal decalcification agents in histopathology:
Rapid action: Should remove calcium quickly without delaying tissue processing.
Preserve tissue morphology: Must not damage or distort cellular and structural details.
Preserve staining qualities: Should not interfere with subsequent staining procedures (H&E, special stains).
Non‑destructive to nucleic acids and proteins: Should allow molecular studies (DNA/RNA/protein analysis) if needed.
Minimal shrinkage or swelling: Should maintain tissue size and architecture.
Easy penetration: Should diffuse uniformly into the tissue for complete decalcification.
Stable and safe: Should be chemically stable, non‑toxic, and easy to handle.
Compatible with fixatives: Should work well with common fixatives like formalin.
Q.14 Write a short note on care and maintenance of the freezing microtome.
Care and Maintenance of Freezing Microtome
Cleaning after use: Always remove tissue debris and frost from the stage and knife holder immediately after sectioning.
Lubrication: Apply a thin layer of suitable lubricant to moving parts regularly to ensure smooth operation.
Knife care: Keep the microtome knife sharp, clean, and free from rust. Store it in a protective case when not in use.
Temperature control: Ensure the freezing stage is maintained at the correct temperature; avoid excessive frost formation.
Avoid corrosion: Wipe down metallic parts to prevent rusting, especially after exposure to moisture.
Calibration: Periodically check and adjust the thickness setting mechanism for accuracy.
Safe handling: Handle the knife and moving parts carefully to prevent accidents and damage.
Storage: Cover the instrument when not in use to protect it from dust and environmental damage.
Q.15 What are the uses of an autopsy?
Uses of Autopsy
Determine cause of death: Establishes the medical reason for death, especially in sudden or unexplained cases.
Study disease processes: Provides valuable information about the progression and effects of diseases on organs and tissues.
Detect medical errors: Helps identify diagnostic mistakes or treatment complications.
Legal and forensic purposes: Assists in criminal investigations, confirming or ruling out foul play.
Public health insights: Contributes to understanding epidemics, new diseases, or environmental hazards.
Medical education and research: Serves as a teaching tool for students and a source of data for scientific studies.
Family reassurance: Offers closure and answers to relatives regarding the cause of death.
Q.16 Write the composition of the Keiserling II solution.
Composition of Kaiserling II Solution
Formalin (37–40% formaldehyde) – 200 mL
Potassium nitrate – 15 g
Potassium acetate – 30 g
Distilled water – up to 1000 mL
Notes
Kaiserling II solution is part of the Kaiserling’s three-solution method for preserving tissues with natural color.
Kaiserling I is the fixative, Kaiserling II is the preservative, and Kaiserling III is the storage solution.
The potassium salts help maintain tissue transparency and color stability.
Q.17 Briefly discuss the FNAC method with a diagram.
FNAC Method – Brief Discussion
Principle: FNAC involves using a fine, hollow needle to aspirate cellular material from a superficial or deep-seated lesion for cytological examination.
Procedure:
A fine needle (usually 22–25 gauge) attached to a syringe is inserted into the lesion.
Negative pressure is applied by pulling back the syringe plunger to aspirate cells.
The material is expelled onto a glass slide, smeared, and stained (commonly with Giemsa or Papanicolaou stain).
Microscopic examination is performed to assess cellular morphology.
Uses: Quick, minimally invasive, cost-effective diagnostic tool for tumors, cysts, lymph nodes, thyroid, breast lumps, etc.
Advantages: Simple, outpatient procedure, avoids surgical biopsy, and rapid diagnosis.
Simple Diagram (conceptual description)
[ Lesion/Mass ]
|
---- Needle ----> Connected to Syringe
|
Aspiration of cells
|
Expelled onto the Glass Slide
|
Smear → Staining → Microscopy
This diagram shows the flow: lesion → needle aspiration → slide preparation → staining → microscopic diagnosis.
Q.18 Write a short note on automation in cytology.
Automation in Cytology
Definition: Automation in cytology refers to the use of advanced instruments, computer-assisted systems, and digital technologies to improve the efficiency, accuracy, and reproducibility of cytological examinations.
Key Features:
Automated slide preparation: Machines prepare smears, fix, and stain slides uniformly.
Automated staining: Ensures consistency in special stains (e.g., Papanicolaou stain).
Image analysis systems: Computer-assisted microscopes scan slides and highlight suspicious cells.
Liquid-based cytology (LBC): Automated systems process samples to reduce artifacts and improve cell preservation.
Digital cytology: High-resolution scanners digitize slides for remote viewing and telepathology.
Advantages:
Reduces human error and variability.
Speeds up processing and reporting.
Improves detection of abnormal cells (e.g., precancerous lesions in cervical cytology).
Facilitates large-scale screening programs.
Limitations:
High cost of equipment.
Requires technical expertise and maintenance.
Still needs human oversight for final diagnosis.
Automation has revolutionized cytology by combining precision, speed, and digital innovation, making diagnostic processes more reliable and scalable.
Q.19 Explain the procedure of a pregnancy test.
A pregnancy test works by detecting the hormone hCG (human chorionic gonadotropin) in urine or blood, which is produced after implantation of a fertilized egg. The test is simple, rapid, and widely used to confirm pregnancy.
Principle
Human Chorionic Gonadotropin (hCG) is secreted by trophoblastic cells of the placenta after implantation.
Pregnancy tests detect hCG either qualitatively (positive/negative) or quantitatively (exact concentration).
hCG can be detected in urine (home test kits) or blood (laboratory tests).
Procedure
1. Urine Pregnancy Test (Home Test Kits)
Collect a fresh urine sample (preferably early morning urine for higher hCG concentration).
Place a few drops on the test strip or dip the strip into the urine.
Wait for 3–5 minutes.
Interpretation:
One line = control (test valid, negative result).
Two lines = control + test line (positive result, hCG present).
2. Laboratory Urine Test
Uses latex particles coated with anti-hCG antibodies.
If hCG is present, agglutination occurs, confirming pregnancy.
3. Blood Test
More sensitive; can detect hCG levels as low as 1 mIU/mL.
Performed in a lab using immunoassays.
Provides quantitative results to monitor pregnancy progression or detect abnormalities.
Key Points
Best time to test: About 1 week after a missed period for reliable results.
False negatives: Can occur if the test is done too early, before hCG levels rise sufficiently.
Clinical use: Besides pregnancy detection, hCG tests are also used in diagnosing ectopic pregnancy, monitoring trophoblastic disease, and certain tumors.
In short, Pregnancy tests detect hCG in urine or blood. Home kits provide quick qualitative results, while lab tests provide more sensitive, quantitative measurements.
Q.20 How will you preserve museum specimens?
Preservation of Museum Specimens
Fixation: Specimens are first fixed in formalin (10% neutral buffered formalin) to prevent autolysis and putrefaction.
Decalcification (if needed): Hard tissues like bone are decalcified using agents such as nitric acid or EDTA.
Kaiserling’s method: A widely used technique for museum specimens. It involves three solutions:
Kaiserling I (formalin, potassium nitrate, potassium acetate) – for fixation.
Kaiserling II – for preservation and maintaining natural color.
Kaiserling III – for long-term storage.
Mounting: Specimens are placed in glass jars filled with preservative solution, ensuring complete immersion.
Labeling: Each specimen is labeled with details like name, diagnosis, and accession number.
Maintenance: Regular checking of fluid levels, topping up with preservative, and replacing cloudy solutions to prevent deterioration.
In short, Museum specimens are preserved by fixation, storage in preservative solutions (such as Kaiserling’s), proper mounting, and regular maintenance to retain their natural appearance and educational value.
Q.21 Explain the process of frozen section cutting.
Specimen Collection
Fresh tissue is obtained during surgery for immediate examination.
The surgeon provides a small tissue sample
No prior fixation is done
Tissue must be kept moist
Freezing the Tissue
The specimen is rapidly frozen to make it firm for sectioning.
Place tissue on the freezing stage of the cryostat
Use liquid CO₂ or refrigerant to freeze
Ensure complete freezing to avoid tearing
Section Cutting
Thin slices of frozen tissue are cut using a freezing microtome/cryostat.
Adjust thickness (usually 5–10 µm)
Cut sections carefully to avoid folds
Transfer sections onto glass slides
Staining the Section
Sections are quickly stained for microscopic examination.
Common stains: Hematoxylin & Eosin (H&E)
Rapid staining protocols were used
Mount with a coverslip
Microscopic Examination
A pathologist examines stained sections to provide an immediate diagnosis.
Check for tumor margins
Identify tissue type and pathology
Report findings to the surgeon
SECTION-D
Note: Long answer type questions. Attempt any two questions out of the three questions. (2x8=16)
Q.23 Give the principle and procedure of the AFB staining method.
Q.24 Describe the principle and procedure of the H & E staining method.
Q.25 “Explain the procedure of autopsy examination.
